Radafaxine
 | |
| Clinical data | |
|---|---|
| ATC code | none |
| Identifiers | |
| |
| CAS Number |
106083-71-0 |
| PubChem (CID) | 9795056 |
| ChemSpider |
7970823 |
| UNII |
Q47741214K |
| ChEMBL |
CHEMBL1172928 |
| Chemical and physical data | |
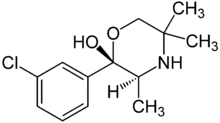
| Formula | C13H18ClNO2 |
| Molar mass | 292.20 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Radafaxine is drug candidate designated GW-353,162[1] by GlaxoSmithKline, investigated for treatment of restless leg syndrome and as an NDRI antidepressant. GlaxoSmithKline was targeting Radafaxine for regulatory filing in 2007,[2] but development was discontinued in 2006 due to "poor test results".[3]
Chemistry
It is a potent metabolite of bupropion, the compound in GlaxoSmithKline's Wellbutrin. More specifically, "hydroxybupropion" is an analogue of bupropion, and radafaxine is an isolated isomer ((2S,3S)-) of hydroxybupropion.[4]
Therefore, radafaxine builds on at least some of the properties of bupropion in humans.[5]
Effects
In various clinical trials, radafaxine has been studied as a treatment for clinical depression, obesity, and neuropathic pain. Radafaxine is described as a norepinephrine-dopamine reuptake inhibitor (NDRI).
Unlike bupropion (which has a much higher effect on dopamine reuptake), radafaxine seems to have a higher potency on norepinephrine. Radafaxine has about 70% of bupropion's efficacy in blocking dopamine reuptake, and 392% of efficacy in blocking norepinephrine reuptake, making it fairly selective for inhibiting the reuptake of norepinephrine over dopamine.[6][7] This, according to GlaxoSmithKline, may account for the increased effect of radafaxine on pain and fatigue.[8]
At least one study suggests that radafaxine has a low abuse potential similar to bupropion.[9]
See also
References
- ↑ Restless Legs Syndrome: First Approval
- ↑ {1} Reviews Novel Therapeutics For CNS Disorders And Confirms Strong Pipeline Momentum - News, Search Jobs, Events
- ↑ Independent.co.uk: GSK breakthrough on bird flu vaccine.
- ↑ radafaxine at the US National Library of Medicine Medical Subject Headings (MeSH)
- ↑ GlaxoSmithKline Reviews Novel Therapeutics for CNS Disorders and Confirms Strong Pipeline Momentum
- ↑ Xu H, Loboz KK, Gross AS, McLachlan AJ (2007). "Stereoselective analysis of hydroxybupropion and application to drug interaction studies". Chirality. 19 (3): 163–70. doi:10.1002/chir.20356. PMID 17167747.
- ↑ Bondarev ML, Bondareva TS, Young R, Glennon RA (2003). "Behavioral and biochemical investigations of bupropion metabolites". Eur J Pharmacol. 474 (1): 85–93. doi:10.1016/S0014-2999(03)02010-7. PMID 12909199.
- ↑ Microsoft PowerPoint - slides_05_burch.ppt
- ↑ Radafaxine