Caroxazone
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | none |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | 18464-39-6 |
| PubChem (CID) | 29083 |
| ChemSpider | 27057 |
| UNII |
807N226MNL |
| ChEMBL | CHEMBL2104164 |
| ECHA InfoCard | 100.038.481 |
| Chemical and physical data | |
| Formula | C10H10N2O3 |
| Molar mass | 206.20 g/mol |
| 3D model (Jmol) | Interactive image |
| |
Caroxazone (Surodil, Timostenil) is an antidepressant which was formerly used for the treatment of depression but is now no longer marketed.[1][2] It acts as a reversible monoamine oxidase inhibitor (RIMA) of both MAO-A and MAO-B subtypes, with five-fold preference for the latter.[3][4][5][6][7]
Synthesis
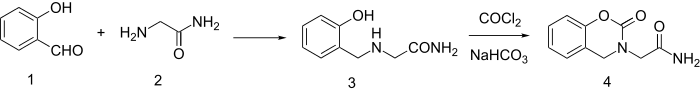
Synthesis starts by reductive amination of salicylaldehyde and glycinamide to give 3. The synthesis is completed by reaction with phosgene and NaHCO3.
See also
References
- ↑ Dictionary of organic compounds. London: Chapman & Hall. 1996. ISBN 0-412-54090-8.
- ↑ Cecchini, S; Petri, P; Ardito, R; Bareggi, S. R.; Torriti, A (1978). "A comparative double-blind trial of the new antidepressant caroxazone and amitriptyline". The Journal of International Medical Research. 6 (5): 388–94. PMID 359383.
- ↑ Monoamine oxidase inhibitors in neurological diseases. New York: M. Dekker. 1994. ISBN 0-8247-9082-0.
- ↑ Moretti, A; Caccia, C; Martini, A; Bonollo, L; Amico, A; Sega, R; Nicolella, V; Nicolis, F. B. (May 1981). "Effect of caroxazone, a new antidepressant drug, on monoamine oxidases in healthy volunteers". British Journal of Clinical Pharmacology. 11 (5): 511–5. doi:10.1111/j.1365-2125.1981.tb01158.x. PMC 1401585
 . PMID 7272163.
. PMID 7272163. - ↑ Moretti, A; Caccia, C; Calderini, G; Menozzi, M; Amico, A (October 1981). "Studies on the mechanism of action of caroxazone, a new antidepressant drug". Biochemical Pharmacology. 30 (19): 2728–31. doi:10.1016/0006-2952(81)90549-9. PMID 6170295.
- ↑ Martini, A; Bonollo, L; Nicolis, F. B.; Sega, R; Palermo, A (June 1981). "Effects of caroxazone, a reversible monoamine oxidase inhibitor, on the pressor response to oral tyramine in man". British Journal of Clinical Pharmacology. 11 (6): 611–5. doi:10.1111/j.1365-2125.1981.tb01178.x. PMC 1402186
 . PMID 7272178.
. PMID 7272178. - ↑ Martini, A; Bonollo, L; Nicolis, F. B.; Sega, R; Palermo, A; Braibanti, E (June 1981). "Effects of caroxazone, a reversible monoamine oxidase inhibitor, on the pressor response to intravenous tyramine in man". British Journal of Clinical Pharmacology. 11 (6): 605–10. doi:10.1111/j.1365-2125.1981.tb01177.x. PMC 1402193
 . PMID 7272177.
. PMID 7272177. - ↑ Bernardi, L; Coda, S; Nicolella, V; Vicario, G. P.; Gioia, B; Minghetti, A; Vigevani, A; Arcamone, F (1979). "Radioisotopic and synthetic studies related to caroxazone metabolism in man". Arzneimittel-Forschung. 29 (9): 1412–6. PMID 583252.
- ↑ Bernardi, L; Coda, S; Pegrassi, L; Suchowsky, G. K. (1968). "Pharmacological properties of some derivatives of 1,3-benzoxazine". Experientia. 24 (8): 774–5. doi:10.1007/bf02144859. PMID 5683159.
- ↑ Bernardi, L; Coda, S; Bonsignori, A; Pegrassi, L; Suchowsky, G. K. (1969). "Central depressant properties of 3,1-benzoxazine derivates". Experientia. 25 (8): 787–8. doi:10.1007/bf01897874. PMID 5348526.
- ↑ GB 1115759; L. Bernardi et al., U.S. Patent 3,427,313 (1965, 1969 both to Soc. Farma. Italia).
This article is issued from Wikipedia - version of the 9/2/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.