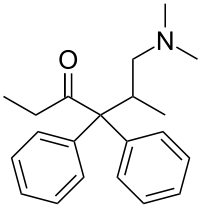
Isomethadone
 | |
| Clinical data | |
|---|---|
| ATC code | None |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| Synonyms | WIN-1783, BW 47-442 |
| CAS Number |
466-40-0 5341-49-1 (HCl) 26594-41-2 ((R)-form) 561-10-4 ((S)-form) 7484-81-2 ((S)-form (HCl)) |
| PubChem (CID) | 10072 |
| ChemSpider | 9675 |
| UNII |
12L95QD6KV |
| Chemical and physical data | |
| Formula | C21H27NO |
| Molar mass | 309.445 g/mol |
| 3D model (Jmol) | Interactive image |
| |
| |
Isomethadone (INN, BAN; Liden), also known as isoamidone, is a synthetic opioid analgesic and antitussive related to methadone that was used formerly as a pharmaceutical drug but is now no longer marketed.[1][2][3][4] Isomethadone was used as both an analgesic and antitussive. It binds to and activates both the μ- and δ-opioid receptors, with the (S)-isomer being the more potent of its two enantiomers.[5] Isomethadone is a Schedule II controlled substance in the United States, with an ACSCN of 9226 and a 2014 aggregate manufacturing quota of 5 grammes. The salts in use are the hydrobromide (free base conversion ratio 0.793), hydrochloride (0.894), and HCl monohydrate (0.850).[6] Isomethadone is also regulated internationally as a Schedule I controlled substance under the United Nations Single Convention on Narcotic Drugs of 1961.[7]
See also
References
- ↑ F.. Macdonald (1997). Dictionary of Pharmacological Agents. CRC Press. p. 1169. ISBN 978-0-412-46630-4. Retrieved 16 May 2012.
- ↑ Dr. Ian Morton; Ian K. M. Morton; Judith M. Hall; Dr. Judith Hall (1999). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer. p. 157. ISBN 978-0-7514-0499-9. Retrieved 16 May 2012.
- ↑ KEATS AS, BEECHER HK (June 1952). "Analgesic potency and side action liability in man of heptazone, WIN 1161-2, 6-methyl dihydromorphine, Metopon, levo-isomethadone and pentobarbital sodium, as a further effort to refine methods of evaluation of analgesic drugs". The Journal of Pharmacology and Experimental Therapeutics. 105 (2): 109–29. PMID 14928215.
- ↑ WINTER CA, FLATAKER L (November 1952). "Antitussive action of d-isomethadone and d-methadone in dogs". Proceedings of the Society for Experimental Biology and Medicine. Society for Experimental Biology and Medicine (New York, N.Y.). 81 (2): 463–5. doi:10.3181/00379727-81-19912. PMID 13027341.
- ↑ Portoghese PS, Poupaert JH, Larson DL, et al. (June 1982). "Synthesis, X-ray crystallographic determination, and opioid activity of erythro-5-methylmethadone enantiomers. Evidence which suggests that mu and delta opioid receptors possess different stereochemical requirements". Journal of Medicinal Chemistry. 25 (6): 684–8. doi:10.1021/jm00348a015. PMID 6284938.
- ↑ http://www.deadiversion.usdoj.gov/fed_regs/quotas/2014/fr0825.htm
- ↑ Thomas Nordegren (1 March 2002). The A-Z Encyclopedia of Alcohol and Drug Abuse. Universal-Publishers. p. 366. ISBN 978-1-58112-404-0. Retrieved 16 May 2012.