Zeisel determination
The Zeisel determination or Zeisel test is a chemical test for the presence of esters or ethers in a chemical substance.[1][2][3][4] It is named after the Czech chemist Simon Zeisel (1854–1933). In a qualitative test a sample is first reacted with a mixture of acetic acid and hydrogen iodide in a test tube. The ensuing reaction results in the cleavage of the ether or the ester into an alkyl iodide and respectively an alcohol or an carboxylic acid.
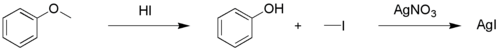
By heating this mixture, the gases are allowed to come into contact with a piece of paper higher up the test tube saturated with mercury(II) nitrate. Any alkyl iodide present will give a reaction with the mercury compound to mercury(II) iodide which has a red or yellow color.
The reaction can also be used to determine the number of methoxy (-OCH3) groups,[5] by distilling the iodomethane into a solution of silver nitrate, which precipitates silver iodide. By filtering and weighing this precipitate it is possible to calculate the number of iodine atoms and hence methoxy groups.
Synthetic applications:
External links
References
- ↑ S Zeisel. Monatshefte für Chemie 6 (1885) p989. http://www.springerlink.com/content/j6n1272v54q05027/
- ↑ S Zeisel. Monatshefte für Chemie 7 (1886) p406. http://www.springerlink.com/content/w135j20507818364/
- ↑ V Prey. Chemische Berichte 74 (1941) p350.
- ↑ Lange. J Org Chem 27 (1962) p2037.
- ↑ Ziesel Reaction