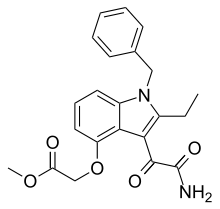
Varespladib methyl
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | none |
| Legal status | |
| Legal status |
|
| Identifiers | |
| Synonyms | A-002 |
| CAS Number |
172733-08-3 |
| PubChem (CID) | 9886917 |
| ChemSpider |
8062590 |
| Chemical and physical data | |
| Formula | C22H22N2O5 |
| Molar mass | 394.4 |
| 3D model (Jmol) | Interactive image |
| |
| |
| | |
Varespladib methyl (also known as A-002, formerly LY333013 and S-3013) is a secretory phospholipase A2 (sPLA2) inhibitor formerly under development by Anthera Pharmaceuticals as a treatment for acute coronary syndrome (ACS).[1] Varespladib methyl is an orally bioavailable prodrug of the molecule varespladib.[2] From 2006 to 2012, varespladib methyl was under active investigation by Anthera Pharmaceuticals as a potential therapy for several inflammatory diseases, including acute coronary syndrome.[3][4] In March 2012, Anthera halted further investigation of varespladib per a recommendation from an independent Data Safety Monitoring Board.[5]
Mechanism
Increased levels of sPLA2 have been observed in patients with cardiovascular disease, and may lead to both acute and chronic disease manifestations by promoting vascular inflammation. Plasma levels of sPLA2 can predict coronary events in patients who recently suffered an ACS as well as in those with stable coronary artery disease.[6][7]
Furthermore, sPLA2 remodels lipoproteins, notably low-density lipoproteins (LDL) and their receptors, which are responsible for removing cholesterol from the body. This remodeling can lead to increased deposition of LDL and cholesterol in the artery wall. In combination with chronic vascular inflammation, these deposits lead to atherosclerosis.[8]
Varespladib inhibits the IIA, V and X isoforms of sPLA2 to reduce inflammation, lower and modulate lipid levels, and reduce levels of C-reactive protein (CRP) and interleukin-6 (IL-6), both indicators of inflammation.[1][9]
History
Varespladib methyl was originally developed jointly by Eli Lilly and Company and Shionogi & Co., Ltd., and was acquired by Anthera Pharmaceuticals in 2006.[10]
A Phase II study demonstrated selective sPLA2 inhibition as well as statistically significant anti-inflammatory responses and reductions in LDL cholesterol levels.[11] Two other Phase II trials, conducted in patients with coronary artery disease, found significant decreases in sPLA2 and LDL cholesterol levels, as well as C-reactive proteins (CRP) and other inflammatory biomarkers.[9][12][13] Varespladib methyl has also been shown to further reduce LDL and inflammatory biomarker levels when administered in conjunction with a cholesterol lowering statin therapy.[14]
In 2010, a Phase III study entitled VISTA-16 was initiated to evaluate the safety and efficacy of short-term treatment with varespladib methyl in subjects with ACS.[15] The trial was halted in March 2012 due to insufficient efficacy.[16] On November 18, 2013, an excess of myocardial infarctions, and of the composite endpoint of cardiovascular mortality, myocardial infarctions and stroke in the VISTA-16 study were reported.[17]
References
- 1 2 "A-002: Short Term (16 week) Treatment of Acute Coronary Syndrome". Anthera Pharmaceuticals, Inc. Retrieved 2011-08-17.
- ↑ Fraser, H., Hislop, C., Christie, R.M., Rick, H.L., Reidy, C.A., Chouinard, M.L., Eacho, P.I., Gould, K.E., Trias, J. (January 2009). "Varespladib (A-002), a secretory phospholipase A2 inhibitor, reduces atherosclerosis and aneurysm formation in ApoE-/- mice.". Journal of Cardiovascular Pharmacology. 53 (1): 60–5. doi:10.1097/FJC.0b013e318195bfbc. PMID 19129734.
- ↑ "Anthera Licenses Portfolio of Anti-Inflammatory Products From Eli Lilly and Company and Shionogi & Co., Ltd." (Press release). Anthera Pharmaceuticals, Inc. 6 September 2006.
- ↑ "Science: sPLA2". Anthera Pharmaceuticals. Retrieved 6 August 2011.
- ↑ "Anthera Halts VISTA-16 Clinical Study Due to Lack of Efficacy Following Recommendation by the Independent Data Safety Monitoring Board" (Press release). Anthera Pharmaceuticals, Inc. 9 March 2012.
- ↑ Mallat, Z., Steg, G., Benessiano, J., Tanguy, M., Fox, K.A., Collet, J., Dabbous, O.H., Henry, P., Carruthers, K.F., Dauphin, A., Arguelles, C.S., Masliah, J., Hugel, B., Montalescot, G., Freyssinet, J., Asselian, B., Tedgui, A. (October 2005). "Circulating Secretory Phospholipase A2 Activity Predicts Recurrent Events in Patients With Severe Acute Coronary Syndromes". Journal of the American College of Cardiology. 46 (7): 1249–57. doi:10.1016/j.jacc.2005.06.056. PMID 16198839.
- ↑ Kugiyama, K., Ota, Y., Takazoe, K., Moriyama, Y., Kawano, H., Miyao, Y., Sakamoto, T., Soejima, H., Ogawa, H., Doi, H., Sugiyama, S., Yasue, H. (September 1999). "Circulating Levels of Secretory Type II Phospholipase A2 Predict Coronary Events in Patients with Coronary Artery Disease". Circulation. 100 (12): 1280–4. doi:10.1161/01.CIR.100.12.1280. PMID 10491371.
- ↑ Mallat, Z., Lambeau, G., Tedgui, A. (November 2010). "Lipoprotein-Associated and Secreted Phospholipases A2 in Cardiovascular Disease: Roles as Biological Effectors and Biomarkers.". Circulation. 122 (21): 2183–200. doi:10.1161/CIRCULATIONAHA.110.936393. PMID 21098459.
- 1 2 Rosenson, R.S., Elliott, M., Stasiv, Y., Hislop, C. (April 2011). "Randomized trial of an inhibitor of secretory phospholipase A2 on atherogenic lipoprotein subclasses in statin-treated patients with coronary heart disease". European Heart Journal. 32 (8): 999–1005. doi:10.1093/eurheartj/ehq374. PMID 21081550.
- ↑ "Anthera Licenses Portfolio of Anti-Inflammatory Products From Eli Lilly and Company and Shionogi & Co., Ltd." (Press release). Anthera Pharmaceuticals, Inc. 6 September 2006.
- ↑ "Anthera's Varespladib Meets Primary Endpoint In Phase 2 Francis Trial For The Treatment Of Acute Coronary Syndrome" (Press release). Anthera Pharmaceuticals, Inc. 6 May 2009.
- ↑ Rosenson R.S.; Hislop C.; McConnell D.; Elliott M; Stasiv Y; Wang N; Waters D.D. (February 2009). "Effects of 1-H-indole-3-glyoxamide (A 002) on concentration of secretory phospholipase A2 (PLASMA study): a phase II double-blind, randomized, placebo-controlled trial". The Lancet. 373 (9664): 649–58. doi:10.1016/S0140-6736(09)60403-7. PMID 19231633.
- ↑ Rosenson, R.S., Fraser, H., Trias, J., Hislop, C. (October 2010). "Varespladib methyl in cardiovascular disease". Expert Opinions in Investigational Drugs. 19 (10): 1245–55. doi:10.1517/13543784.2010.517193. PMID 20809869.
- ↑ Rosenson, R.S., Hislop, C., Elliott, M., Stasiv, Y., Goulder, M., Waters, D. (September 2010). "Effects of varespladib methyl on biomarkers and major cardiovascular events in acute coronary syndrome patients". Journal of American College of Cardiology. 56 (14): 1079–88. doi:10.1016/j.jacc.2010.06.015. PMID 20863951.
- ↑ "Anthera Enrolls First Patients in Pivotal Varespladib Phase 3 Clinical Study" (Press release). Anthera Pharmaceuticals, Inc. 23 June 2010.
- ↑ ClinicalTrials.gov. "VISTA-16 Trial: Evaluation of Safety and Efficacy of Short-term A-002 Treatment in Subjects With Acute Coronary Syndrome.". United States National Institute of Health. Retrieved 2011-08-17.
- ↑ Nicholls Stephen J., Kastelein John J. P., Schwartz Gregory G., Bash Dianna, Rosenson Robert S., Cavender Matthew A., Brennan Danielle M., Koenig Wolfgang, Jukema J. Wouter, Nambi Vijay, Wright R. Scott, Menon Venu, Lincoff A. Michael, Nissen Steven E. (2014). "Varespladib and Cardiovascular Events in Patients With an Acute Coronary Syndrome". JAMA. 311 (3): 252. doi:10.1001/jama.2013.282836.