Isothiazole
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
1,2-thiazole | |||
| Other names
isothiazole | |||
| Identifiers | |||
| 288-16-4 | |||
| 3D model (Jmol) | Interactive image | ||
| ChEBI | CHEBI:35600 | ||
| ChemSpider | 60838 | ||
| PubChem | 67515 | ||
| |||
| |||
| Properties | |||
| C3H3NS | |||
| Molar mass | 85.12 g·mol−1 | ||
| Boiling point | 114 °C (237 °F; 387 K)[1] | ||
| Acidity (pKa) | -0.5 (of conjugate acid) [2] | ||
| Related compounds | |||
| Related compounds |
thiazole, isoxazole | ||
| Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| | |||
| Infobox references | |||
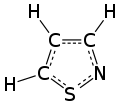
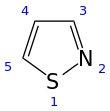
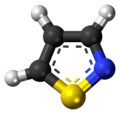
An isothiazole, or 1,2-thiazole, is a type of organic compound containing a five-membered aromatic ring that consists of three carbon atoms, one nitrogen atom, and one sulfur atom.[3] Isothiazole is a member of a class of compounds known as azoles. In contrast to the isomeric thiazole, the two heteroatoms are in adjacent positions.
The ring structure of isothiazole is incorporated into larger compounds with biological activity such as the pharmaceutical drugs ziprasidone and perospirone.
References
- ↑ Isothiazoles, D. W. Brown and M. Sainsbury, page 513
- ↑ Zoltewicz, J. A. & Deady, L. W. Quaternization of heteroaromatic compounds. Quantitative aspects. Adv. Heterocycl. Chem. 22, 71-121 (1978).
- ↑ Heterocyclic Chemistry, 3rd Edition, J.A. Joule, K. Mills, and G.F. Smith, page 394
This article is issued from Wikipedia - version of the 2/9/2016. The text is available under the Creative Commons Attribution/Share Alike but additional terms may apply for the media files.