Glymphatic system
| Glymphatic System | |
|---|---|
|
Mammalian glymphatic system | |
The glymphatic system (or glymphatic clearance pathway) is a functional waste clearance pathway for the vertebrate central nervous system (CNS). The pathway consists of a para-arterial influx route for cerebrospinal fluid (CSF) to enter the brain parenchyma, coupled to a clearance mechanism for the removal of interstitial fluid (ISF) and extracellular solutes from the interstitial compartments of the brain and spinal cord. Exchange of solutes between the CSF and the ISF is driven by arterial pulsation and regulated during sleep by the expansion and contraction of brain extracellular space. Clearance of soluble proteins, waste products, and excess extracellular fluid is accomplished through convective bulk flow of the ISF, facilitated by/ astrocytic aquaporin 4 (AQP4) water channels.
The name "glymphatic system" was coined by the Danish neuroscientist Maiken Nedergaard in recognition of its dependence upon glial cells and the similarity of its functions to those of the peripheral lymphatic system.[1]
While glymphatic flow was initially believed to be the complete answer to the long-standing question of how the sensitive neural tissue of the CNS functions in the perceived absence of a lymphatic drainage pathway for extracellular proteins, excess fluid, and metabolic waste products, two subsequent articles by Louveau et al. from the University of Virginia School of Medicine and Aspelund et al. from the University of Helsinki reported independently the discovery that the dural sinuses and meningeal arteries are in fact lined with conventional lymphatic vessels, and that this long-elusive vasculature forms the connecting pathway for the entrance and exit of lymphatic fluid and immune cells from the meningeal compartment to the glymphatic system.[2][3]
Background
Description of the cerebrospinal fluid
Although the first known observations of the CSF date back to Hippocrates (460–375 BCE) and later Galen (130–200 CE), its discovery is credited to Emanuel Swedenborg (1688–1772 CE), who, being a devoutly religious man, identified the CSF during his search for the seat of the soul.[4] The 16 centuries of anatomists that came after Hippocrates and Galen may have missed identifying the CSF due to the time period's prevailing autopsy technique, which included severing the head and draining the blood before dissecting the brain.[4] Although Swedenborg's work (in translation) was not published until 1887 due to his lack of medical credentials, he may have also made the first connection between the CSF and the lymphatic system. His description of the CSF was of a "spirituous lymph".[4]
Absence of CNS lymphatics
In the peripheral organs, the lymphatic system performs important immune functions, and runs parallel to the blood circulatory system to provide a secondary circulation that transports excess interstitial fluid, proteins and metabolic waste products from the systemic tissues back into the blood. The efficient removal of soluble proteins from the interstitial fluid is critical to the regulation of both colloidal osmotic pressure and homeostatic regulation of the body's fluid volume. The importance of lymphatic flow is especially evident when the lymphatic system becomes obstructed. In lymphatic associated diseases such as elephantiasis (where parasites occupying the lymphatic vessels block the flow of lymph), the impact of such an obstruction can be dramatic. The resulting chronic edema is due to the breakdown of lymphatic clearance and the accumulation of interstitial solutes. In 2015, the presence of a cerebral lymphatic system was first identified by two independent studies by Louveau et al. and Aspelund et al.[2][3] overturning long held dogma that the CNS does not contain a lymphatic system.
Diffusion hypothesis
For over a century the prevailing hypothesis was that the flow of cerebrospinal fluid (CSF), which surrounds but does not come in direct contact with the parenchyma of the CNS, could supplant peripheral lymphatic functions and play an important role in the clearance of extracellular solutes. The majority of the CSF is formed in the choroid plexus and flows through the brain along a distinct pathway: moving through the cerebral ventricular system, into the subarachnoid space surrounding the brain, then draining into the systemic blood column via arachnoid granulations of the dural sinuses or to peripheral lymphatics along cranial nerve sheathes.[5][6] Many researchers have suggested that the CSF compartment constitutes a sink for interstitial solute and fluid clearance from the brain parenchyma. However, the distances between the interstitial fluid and the CSF in the ventricles and subarachnoid space are too great for the efficient removal of interstitial macromolecules and wastes by simple diffusion alone. Helen Cserr at Brown University calculated that mean diffusion times for large molecules such as albumin would exceed 100hrs to traverse 1 cm of brain tissue,[7] a rate that is not compatible with the intense metabolic demands of brain tissue. A clearance system based on simple diffusion would additionally lack the sensitivity to respond rapidly to deviations from homeostatic conditions.
Key determinants of diffusion through the brain interstitial spaces are the dimensions and composition of the extracellular compartment. In a series of elegantly designed experiments in the 1980s and 1990s, C. Nicholson and colleagues from New York University explored the microenvironment of the extracellular space using ion-selective micropipettes and ionophoretic point sources. Using these techniques Nicholson showed that solute and water movement through the brain parenchyma slows as the extracellular volume fraction decreases and becomes more tortuous.[8]
As an alternative explanation to diffusion, Cserr and colleagues proposed that convective bulk flow of interstitial fluid from the brain parenchyma to the CSF was responsible for efficient waste clearance[7]
Progress in the field of CSF dynamics
Experiments conducted at the University of Maryland in the 1980s by Patricia Grady and colleagues postulated the existence of solute exchange between the interstitial fluid of the brain parenchyma and the CSF via paravascular spaces. In 1985, Grady and colleagues suggested that cerebrospinal fluid and interstitial fluid exchange along specific anatomical pathways within the brain, with CSF moving into the brain along the outside of blood vessels. Grady's group suggested that these 'paravascular channels' were functionally analogous to peripheral lymph vessels, facilitating the clearance of interstitial wastes from the brain.[9][10] Other labs at the time, however, did not observe such widespread paravascular CSF–ISF exchange.[7][11][12][13]
The continuity between the brain interstitial fluid and the CSF was confirmed by H. Cserr and colleagues from Brown University and Kings College London.[13] The same group postulated that interstitial solutes in the brain parenchyma exchange with CSF via a bulk flow mechanism, rather than diffusion. However other work from this same lab indicated that the exchange of CSF with interstitial fluid was inconsistent and minor, contradicting the findings of Grady and colleagues.[11][12]
Modern model of organ wide clearance
Key features
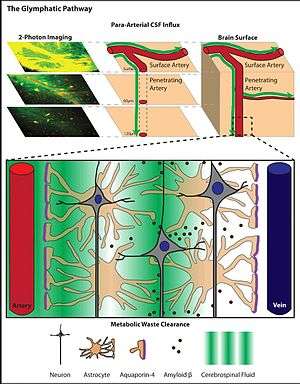
In a study published in 2012,[14] a group of researchers from the University of Rochester headed by M. Nedergaard used in-vivo two-photon imaging of small fluorescent tracers to monitor the flow of subarachnoid CSF into and through the brain parenchyma. The two-photon microscopy allowed the Rochester team to visualize the flux of CSF in living mice, in real time, without needing to puncture the CSF compartment (imaging was performed through a closed cranial window). According to findings of that study, subarachnoid CSF enters the brain rapidly, along the paravascular spaces surrounding the penetrating arteries, then exchanges with the surrounding interstitial fluid.[14] Similarly, interstitial fluid was cleared from the brain parenchyma via the paravascular spaces surrounding large draining veins.
| Glymphatic System | |
|---|---|
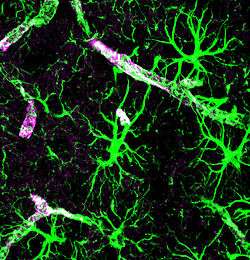 Astrocytes stained for GFAP (green) and aquaporin-4 (purple) | |
Paravascular spaces are CSF-filled channels formed between the brain blood vessels and leptomeningeal sheathes that surround cerebral surface vessels and proximal penetrating vessels. Around these penetrating vessels, paravascular spaces take the form of Virchow-Robin spaces. Where the Virchow-Robin spaces terminate within the brain parenchyma, paravascular CSF can continue traveling along the basement membranes surrounding arterial vascular smooth muscle, to reach the basal lamina surrounding brain capillaries. CSF movement along these paravascular pathways is rapid and arterial pulsation has long been suspected as an important driving force for paravascular fluid movement.[9] In a study published in 2013, J. Iliff and colleagues demonstrated this directly. Using in vivo 2-photon microscopy, the authors reported that when cerebral arterial pulsation was either increased or decreased, the rate of paravacular CSF flux in turn increased or decreased, respectively.
Astrocytes extend long processes that interface with neuronal synapses, as well as projections referred to as 'end-feet' that completely ensheathe the brain's entire vasculature. Although the exact mechanism is not completely understood, astrocytes are known to facilitate changes in blood flow[15][16] and have long been thought to play a role in waste removal in the brain.[17] Researchers have long known that astrocytes express water channels called aquaporins.[18] Until recently, however, no physiological function has been identified that explains their presence in the astrocytes of the mammalian CNS. Aquaporins are membrane-bound channels that play critical roles in regulating the flux of water into and out of cells. Relative to simple diffusion, the presence of aquaporins in biological membranes facilitates a 3–10 fold increase in water permeability.[19] Two types of aquaporins are expressed in the CNS: aquaporin-1, which is expressed by specialized epithelial cells of the choroid plexus, and aquaporin-4 (AQP4), which is expressed by astrocytes.[20][21] Aquaporin-4 expression in astrocytes is highly polarized to the endfoot processes ensheathing the cerebral vasculature. Up to 50% of the vessel-facing endfoot surface that faces the vasculature is occupied by orthogonal arrays of AQP4.[18][20] In 2012, it was shown that AQP4 is essential for paravascular CSF–ISF exchange. Analysis of genetically modified mice that lacked the AQP4 gene revealed that the bulk flow-dependent clearance of interstitial solutes decreases by 70% in the absence of AQP4. Based upon this role of AQP4-dependent glial water transport in the process of paravascular interstitial solute clearance, Iliff and Nedergaard termed this brain-wide glio-vascular pathway the 'glymphatic system'.
Physiological functions
Increased waste clearance during sleep
A publication by L. Xie and colleagues in 2013 explored the efficiency of the glymphatic system during slow wave sleep and provided the first direct evidence that the clearance of interstitial waste products increases during the resting state. Using a combination of diffusion ionophoresis techniques pioneered by Nicholson and colleagues, in vivo 2-photon imaging, and electroencephalography to confirm the wake and sleep states, Xia and Nedergaard demonstrated that the changes in efficiency of CSF–ISF exchange between the awake and sleeping brain were caused by expansion and contraction of the extracellular space, which increased by ~60% in the sleeping brain to promote clearance of interstitial wastes such as amyloid beta.[22] On the basis of these findings, they hypothesized that the restorative properties of sleep may be linked to increased glymphatic clearance of metabolic waste products produced by neural activity in the awake brain.
Transport of lipids
Another key function of the glymphatic system was documented by Thrane et al., who in 2013 demonstrated that the brain's system of paravascular pathways plays an important role in transporting small lipophilic molecules.[23] Led by M. Nedergaard, Thrane and colleagues also showed that the paravascular transport of lipids through the glymphatic pathway activated glial calcium signalling and that the depressurization of the cranial cavity, and thus impairment of the glymphatic circulation, led to unselective lipid diffusion, intracellular lipid accumulation and pathological signalling among astrocytes. Although further experiments are needed to parse out the physiological significance of the connection between the glymphatic circulation, calcium signalling and paravascular lipid transport in the brain, the findings point to the adoption of a function in the CNS similar to the capacity of the intestinal lymph vessels (lacteals) to carry lipids to the liver.
Implications for neurological disease
Pathologically, neurodegenerative diseases such as amyotrophic lateral sclerosis, Alzheimer's disease, Parkinson's disease and Huntington's disease are all characterized by the progressive loss of neurons, cognitive decline, motor impairments, and sensory loss.[24][25] Collectively these diseases fall within a broad category referred to as proteinopathies, due to the common assemblage of misfolded or aggregated intracellular or extracellular proteins. According to the prevailing amyloid hypothesis of Alzheimer's disease, the aggregation of amyloid-beta (a peptide normally produced in and cleared from the healthy young brain) into extracellular plaques drives the neuronal loss and brain atrophy that is the hallmark of Alzheimer's dementia. Although the full extent of the glymphatic system's involvement in Alzheimer's disease and other neurodegenerative disorders remains unclear, researchers have demonstrated through experiments with genetically modified mice that the proper function of the glymphatic clearance system was necessary to remove soluble amyloid-beta from the brain interstitium.[14] In mice that lack the AQP4 gene, amyloid-beta clearance is reduced by approximately 55 percent.
The glymphatic system may also be impaired after acute brain injuries such as ischemic stroke, intracranial hemorrhage or subarachnoid hemorrhage. In 2014, a group of researchers from the French Institute of Health and Medical Research (INSERM) demonstrated by MRI that the glymphatic system was impaired after subarachnoid hemorrhage, because of the presence of coagulated blood in the paravascular spaces.[26] Interestingly, injection of tissue plasminogen activator (a fibrinolytic drug) in the CSF improved glymphatic functioning. In a parallel study, they also demonstrated that the glymphatic system was impaired after ischemic stroke in the ischemic hemisphere, although the pathophysiological basis of this phenomenon remains unclear. Notably, recanalization of the occluded artery also reestablished the glymphatic flow.
Further reading
- Konnikova, Maria (2014-01-11). "Goodnight. Sleep Clean.". New York Times. Retrieved 2014-01-20.
- Shaw, Gina (2015-07-10). "New Study Suggests Brain Is Connected to the Lymphatic System: What the Discovery Could Mean for Neurology". AAN. Retrieved 2015-07-10.
References
- ↑ Konnikova, Maria (11 January 2014). "Goodnight. Sleep Clean.". The New York Times. Retrieved 18 February 2014.
She called it the glymphatic system, a nod to its dependence on glial cells
- 1 2 Antoine Louveau, Igor Smirnov, Timothy J. Keyes, Jacob D. Eccles, Sherin J. Rouhani, J. David Peske, Noel C. Derecki, David Castle, James W. Mandell, Kevin S. Lee, Tajie H. Harris, Jonathan Kipnis. (2015). "Structural and functional features of central nervous system lymphatic vessels.". Nature. 523: 337–41. doi:10.1038/nature14432. PMID 26030524.
- 1 2 Aleksanteri Aspelund, Salli Antila, Steven T. Proulx, Tine Veronica Karlsen, Sinem Karaman, Michael Detmar, Helge Wiig, Kari Alitalo. (2015). "A dural lymphatic vascular system that drains brain interstitial fluid and macromolecules.". Journal of Experimental Medicine. 212: 991–9. doi:10.1084/jem.20142290. PMID 26077718.
- 1 2 3 Hajdu, Steven (2003). "A Note from History: Discovery of the Cerebrospinal Fluid" (PDF). Annals of Clinical and Laboratory Science. 33 (3).
- ↑ Abbott NJ (2004). "Evidence for bulk flow of brain interstitial fluid: significance for physiology and pathology". Neurochem Int. 45 (4): 545–52. doi:10.1016/j.neuint.2003.11.006. PMID 15186921.
- ↑ Bradbury MW, Cserr HF, Westrop RJ (1981). "Drainage of cerebral interstitial fluid into deep cervical lymph of the rabbit". Am J Physiol. 240 (4): F329–36. PMID 7223890.
- 1 2 3 Cserr HF (1971). "Physiology of the choroid plexus". Physiol Rev. 51 (2): 273–311. PMID 4930496.
- ↑ C. Nicholson, J. M. Phillips (1981). "Ion diffusion modified by tortuosity and volume fraction in the extracellular microenvironment of the rat cerebellum". The Journal of Physiology. 321 (1): 225–257. doi:10.1113/jphysiol.1981.sp013981. PMID 7338810. Retrieved 9 December 2013.
- 1 2 Rennels ML, Gregory TF, Blaumanis OR, Fujimoto K, Grady PA (1985). "Evidence for a 'paravascular' fluid circulation in the mammalian central nervous system, provided by the rapid distribution of tracer protein throughout the brain from the subarachnoid space". Brain Res. 326 (1): 47–63. doi:10.1016/0006-8993(85)91383-6. PMID 3971148.
- ↑ Rennels ML, Blaumanis OR, Grady PA (1990). "Rapid solute transport throughout the brain via paravascular fluid pathways". Adv Neurol. 52: 431–9. PMID 2396537.
- 1 2 Pullen RG, DePasquale M, Cserr HF (1987). "Bulk flow of cerebrospinal fluid into brain in response to acute hyperosmolality". Am J Physiol. 253 (3 Pt 2): F538–45. PMID 3115117.
- 1 2 Ichimura T, Fraser PA, Cserr HF (1991). "Distribution of extracellular tracers in perivascular spaces of the rat brain". Brain Res. 545 (1–2): 103–13. doi:10.1016/0006-8993(91)91275-6. PMID 1713524.
- 1 2 Cserr HF, Cooper DN, Suri PK, Patlak CS (1981). "Efflux of radiolabeled polyethylene glycols and albumin from rat brain". Am J Physiol. 240 (4): F319–28. PMID 7223889.
- 1 2 3 Iliff JJ, Wang M, Liao Y, Plogg BA, Peng W, Gundersen GA, Benveniste H, Vates GE, Deane R, Goldman SA, Nagelhus EA, Nedergaard M (2012). "A Paravascular Pathway Facilitates CSF Flow Through the Brain Parenchyma and the Clearance of Interstitial Solutes, Including Amyloid β". Sci Trans Med. 4 (147): 147ra111. doi:10.1126/scitranslmed.3003748. PMC 3551275
 . PMID 22896675.
. PMID 22896675. - ↑ Takano T, Tian GF, Peng W, Lou N, Libionka W, Han X, Nedergaard M (2006). "Astrocyte-mediated control of cerebral blood flow". Nat Neurosci. 9 (2): 260–7. doi:10.1038/nn1623. PMID 16388306.
- ↑ Schummers J, Yu H, Sur M (2008). "Tuned Responses of Astrocytes and Their Influence on Hemodynamic Signals in the Visual Cortex". Science. 320 (5883): 1638–43. doi:10.1126/science.1156120. PMID 18566287.
- ↑ Yuhas, Daisy (2012). "How the brain cleans itself". Scientific American.
- 1 2 Amiry-Moghaddam M, Ottersen OP (2003). "The molecular basis of water transport in the brain". Nature Reviews Neuroscience. 4 (12): 991–1001. doi:10.1038/nrn1252. PMID 14682361.
- ↑ Verkman AS, Mitra AK (2000). "Structure and function of aquaporin water channels". Am J Physiol Renal Physiol. 278 (1): F13–28. PMID 10644652.
- 1 2 Verkman AS, Binder DK, Bloch O, Auguste K, Papadopoulos MC (2006). "Three distinct roles of aquaporin-4 in brain function revealed by knockout mice". Biochim Biophys Acta. 1758 (8): 1085–93. doi:10.1016/j.bbamem.2006.02.018. PMID 16564496.
- ↑ Yool AJ (2007). "Aquaporins: multiple roles in the central nervous system". Neuroscientist. 13 (5): 470–85. doi:10.1177/1073858407303081. PMID 17901256.
- ↑ Lulu Xie, Hongyi Kang1, Qiwu Xu, Michael J. Chen, Yonghong Liao, Meenakshisundaram Thiyagarajan, John O'Donne, Daniel J. Christensen, Charles Nicholson, Jeffrey J. Iliff, Takahiro Takano, Rashid Deane, Maiken Nedergaard (2013). "Sleep Drives Metabolite Clearance from the Adult Brain". Science. 342 (6156): 373–377. doi:10.1126/science.1241224. PMID 24136970. Retrieved 18 October 2013.
- ↑ Vinita Rangroo Thrane, Alexander S. Thrane, Benjamin A. Plog, Meenakshisundaram Thiyagarajan, Jeffrey J. Iliff, Rashid Deane, Erlend A. Nagelhus, Maiken Nedergaard (2013). "Paravascular microcirculation facilitates rapid lipid transport and astrocyte signaling in the brain". Scientific Reports. 3 (2582). doi:10.1038/srep02582. Retrieved 9 December 2013.
- ↑ Mehler MF, Gokhan S (2000). "Mechanisms underlying neural cell death in neurodegenerative diseases: alterations of a developmentally-mediated cellular rheostat". Trends Neurosci. 23 (12): 599–605. doi:10.1016/s0166-2236(00)01705-7. PMID 11137149.
- ↑ Narasimhan, Kalyani (2006). "Quantifying motor neuron loss in ALS". Nature Neuroscience.
- ↑ Gauberti, Gaberel (2014). "Impaired Glymphatic Perfusion After Strokes Revealed by Contrast-Enhanced MRI: A New Target for Fibrinolysis?". Stroke.