Aniline acetate test
| Classification | Colorimetric method |
|---|---|
| Analytes | Pentoses |
The aniline acetate test is a chemical test to identify the presence of certain carbohydrates. These carbohydrates may be converted (by hydrochloric acid) to furfural, which reacts with aniline acetate to produce a bright pink color.
Procedure and mechanism
A dry sample is dissolved in a small volume of hydrochloric acid and briefly heated. A piece of paper, previously impregnated with aniline acetate, is exposed to the vapor from the sample solution. A bright pink color on the paper is positive for the presence of pentoses.
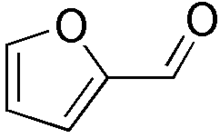
Hydrochloric acid dehydrates pentoses (sugars containing five carbon atoms) to produce furfural. The reaction of furfural and aniline produces a bright pink color. Hexoses, which are sugars which contain six carbons, are not dehydrated to furfural, and so they do not produce a pink color.
Interferences
3-Furanaldehyde responds to the usual tests for aldehydes, but unlike 2-furanaldehyde it gives no color test with aniline acetate.
References
- A Method for the identification of pure organic compounds by a systematic analytical procedure based on physical properties and chemical reactions. v. 1, 1911. By Samuel Parsons Mulliken. Published by J. Wiley & Sons, Inc., 1904. Google Books link page 33.
- "Modifications of the aniline acetate-furfural method for the determination of pentose." The Analyst, 1956, 81, 598 - 601, doi:10.1039/AN9568100598